Attention Deficit Disorder Prosthetic Memory Program
Flash freezing
ADDPMP678
In physics and chemistry, flash freezing is the process whereby objects are frozen in just a few hours by subjecting them to cryogenic temperatures, or through direct contact with liquid nitrogen at −196 °C (−320.8 °F).
Flash freezing is commonly used in the food industry to quickly freeze perishable food items. In this case, food items are subjected to temperatures well below water’s melting/freezing point. Thus, smaller ice crystals are formed, causing less damage to cell membranes.
Flash freezing techniques are also used to freeze biological samples quickly so that large ice crystals cannot form and damage the sample. This rapid freezing is done by submerging the sample in liquid nitrogen or a mixture of dry ice and ethanol.




ADDPMP237
Tangerine Dream Equipment
ADDPMP543
Ribs Recordings

ADDPMP772
Shimizu Mega-City

ADDPMP069
Elementi di semiologia

ADDPMP402
Tropical Cyclone Naming

ADDPMP570
La Guardia Committee
ADDPMP203
Tamu Massif

ADDPMP118
Parco dei Mostri

ADDPMP511
Controlled Impact Demonstration

ADDPMP112
Anthony Braxton's Composition 113

ADDPMP134
Faraday Cage

ADDPMP053
The Planetary Society
ADDPMP001
Beach Portraits

ADDPMP002
About Nothing (1992)

ADDPMP003
3PM

ADDPMP004
Alexis Zorbas (1964)

ADDPMP005
The Urantia Book

ADDPMP006
Best Cry Ever

ADDPMP007
Brave New World

ADDPMP008
Harness

ADDPMP009
Columbine Shooters

ADDPMP010
Propaganda Loudspeakers

ADDPMP011
Emotional Support Animal

ADDPMP012
Hannibal Lecter’s Memory Palace

ADDPMP013
Witching Hour

ADDPMP014
Challenger Deep
ADDPMP015
Luther Blissett Collective

ADDPMP016
Magnus Hirschfeld

ADDPMP017
Objectophilia

ADDPMP018
Kuwait Water Towers

ADDPMP019
Quantum Zeno Effect
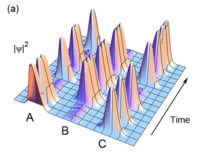
ADDPMP020
Razzle Dazzle Camouflage

ADDPMP021
Cerne Abbas Giant

ADDPMP022
Sea Org Contract

ADDPMP023
Self-Hypnosis

ADDPMP024
Sirius (1977)

ADDPMP025
SIS Building

ADDPMP026
Improvised Weapon
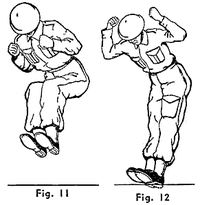
ADDPMP027
The Fourth Sex

ADDPMP028
Telephone drawing

ADDPMP029
Sea Shadow (IX-529)

ADDPMP030
O.J. Simpson Bronco Chase

ADDPMP031
Empire

ADDPMP032
The Blair Witch Project (1999)

ADDPMP033
Gabrielle d'Estrées

ADDPMP034
Poison Dart frog

ADDPMP035
Myanmar Coup Aerobics

ADDPMP036
Stadium Effect

ADDPMP037
Hypersomnia
ADDPMP038
The Sublime

ADDPMP039
Baikonur Cosmodrome

ADDPMP040
The Damned Cast Into Hell

ADDPMP041
Da Vinci

ADDPMP042
Olmec Colossal Heads

ADDPMP043
RLA

ADDPMP044
Acoustic Mirrors

ADDPMP045
Swiss Rebels

ADDPMP046
Hidden Peak

ADDPMP047
Videodrome

ADDPMP048
Dictators Interior Architecture

ADDPMP049
Absorbance
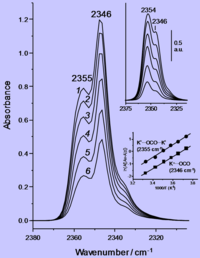
ADDPMP050
Hypernormalization

ADDPMP051
Wound Man
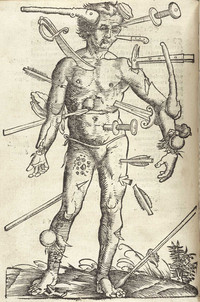
ADDPMP052
Ramses II Passeport

ADDPMP053
The Planetary Society

ADDPMP054
Boomtown

ADDPMP055
Peak Ground Acceleration

ADDPMP056
Tales of Mystery and Imagination

ADDPMP057
Burning Piano

ADDPMP058
Ryugyong

ADDPMP059
Lowdown Da Sinista

ADDPMP060
Rogue Waves
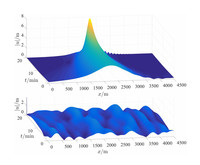
ADDPMP061
Toshio Saeki

ADDPMP062
Kukeri

ADDPMP063
Laser Pointer

ADDPMP064
Binaural Recording

ADDPMP065
Armoured Presidential Limousine

ADDPMP066
Gynecological Gymnastics from Outer Space

ADDPMP067
Broken Noses

ADDPMP068
Drone Hunting Eagles

ADDPMP069
Elementi di semiologia

ADDPMP070
The emergence of space
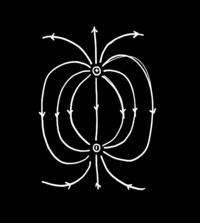
ADDPMP071
Mundaneum
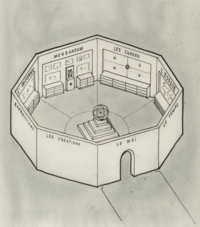
ADDPMP072
20 Shot Sequence

ADDPMP073
The Anthropocene Era

ADDPMP074
Séance de Travail

ADDPMP075
Vapor Cone

ADDPMP076
Soong Ching Ling monument

ADDPMP077
Suck

ADDPMP078
Chinese military parades

ADDPMP079
Fingerprint Verification

ADDPMP080
Ato boldon

ADDPMP081
309th Aerospace Maintenance and Regeneration Group

ADDPMP082
Axis of Evil
ADDPMP083
hybrid creatures

ADDPMP084
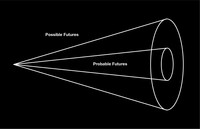
The cone of plausibility
ADDPMP085
4'33"

ADDPMP086
Dis-Armor by Krzysztof Wodiczko

ADDPMP087
Doom Controversy

ADDPMP088
Conquistadors of the Useless

ADDPMP089
Peter Cain's Cars painting series

ADDPMP090
Muammar Gaddafi's Taylored Ensembles

ADDPMP091
Remote Control Beetles

ADDPMP092
Cité Pablo Picasso

ADDPMP093
Overview effect

ADDPMP094
Sam Little Portraits

ADDPMP095
NFL Banned Facemasks

ADDPMP096
Saya Hnin-Mahla

ADDPMP097
Osama Bin Laden's Video Collection

ADDPMP098
Metal Melting Point Chart

ADDPMP099
Dictionary of the Underworld

ADDPMP100
Tameshiwari

ADDPMP101
Ulvetanna

ADDPMP102
Jouko Lehtola

ADDPMP103
Césarée by Marguerite Duras

ADDPMP104
Snakebot

ADDPMP105
Optical Camouflage

ADDPMP106
Sheikh Hamad bin Hamdan Al Nahyan

ADDPMP107
Balkan sworn virgins

ADDPMP108
Punk Zines

ADDPMP109
Millwall brick
ADDPMP110
Let's Take Back Our Space

ADDPMP111
Low Orbital Spider Webs

ADDPMP112
Anthony Braxton's Composition 113
ADDPMP113
San Siro

ADDPMP114
Laurent Melki

ADDPMP115
Systema Naturae

ADDPMP116
GAINSBOURG BURNING 500 FRANCS

ADDPMP117
Chaturbate Empty Rooms

ADDPMP118
Parco dei Mostri

ADDPMP119
Atlantis
ADDPMP120
St. Elmo's Fire

ADDPMP121
Electron Micrograph Portraits

ADDPMP122
Den

ADDPMP123
Colors Of Noise
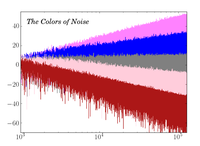
ADDPMP124
Tooth Universal Numbering System
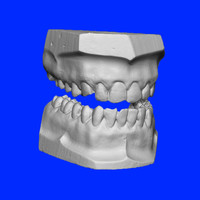
ADDPMP125
Eye Tracking
ADDPMP126
Katherine Johnson
ADDPMP127
Gold To Go

ADDPMP128
Acéphale

ADDPMP129
Cadillac Ranch

ADDPMP130
Polyphemus

ADDPMP131
The Music of the Spheres

ADDPMP132
Pollination

ADDPMP133
Prison Map
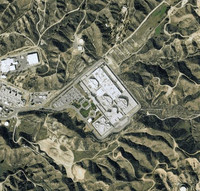
ADDPMP134
Faraday Cage

ADDPMP135
Dennis Tito

ADDPMP136
Code of Hammurabi

ADDPMP137
Hoverbike

ADDPMP138
Attacus Atlas

ADDPMP139
Barras Bravas

ADDPMP140
We Can't Be Stopped Album Cover

ADDPMP141
Project Grizzly

ADDPMP142
Hydra-Shok

ADDPMP143
Pickpocket

ADDPMP144
Roberto Carlos Impossible Goal Against France

ADDPMP145
Zodiak Free Arts Lab

ADDPMP146
The Mirror Test

ADDPMP147
Machine Learning

ADDPMP148
800 Views Airport

ADDPMP149
Cape Town To Magadan
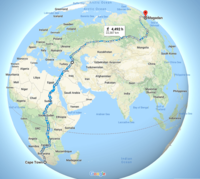
ADDPMP150
ROOM 666

ADDPMP151
Nasa Aerogel

ADDPMP152
The World Saves Abu Simbel

ADDPMP153
Ophthalmic Workstations Design

ADDPMP154
Transcendence
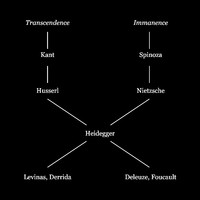
ADDPMP155
Swarm Behaviour

ADDPMP156
The Psychiatric Bulletin
ADDPMP157
Inferno Planet Wasp-76b

ADDPMP158
Scaled Composites Model 351 Stratolaunch

ADDPMP159
Me At The Zoo

ADDPMP160
Illegal Number

ADDPMP161
Hoover Dam Flag

ADDPMP162
Minecraft Architecture

ADDPMP163
Ball Lightning

ADDPMP164
In My Room : Teenagers in Their Bedrooms

ADDPMP165
Extraterrestrial Real Estate

ADDPMP166
Nick Gleis Private Jets Interiors

ADDPMP167
Miracle Lone Pine Tree

ADDPMP168
Arpanet
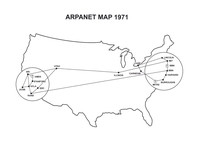
ADDPMP169
Number Of The Beast

ADDPMP170
Largest Hiking Boot

ADDPMP171
Halley Research Station

ADDPMP172
On The Art Of The Cinema

ADDPMP173
Seattle Kingdome Demolition

ADDPMP174
Ancient Egyptian Units of Measurement
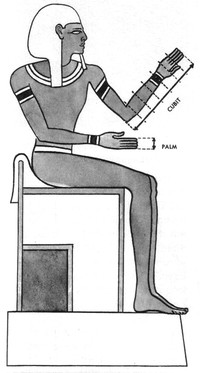
ADDPMP175
DJ Screw Merorablia

ADDPMP176
National Redoubt

ADDPMP177
Oro by Cizia Zykë

ADDPMP178
Shape of the universe
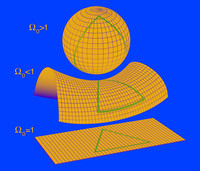
ADDPMP179
Flail

ADDPMP180
Parioscorpio Venator
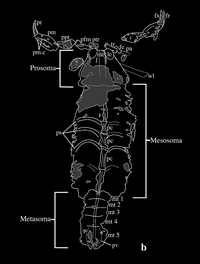
ADDPMP181
14th Street by Wolfgang Tillmans
ADDPMP182
Burden Of Dreams

ADDPMP183
Air-Gap Flash
ADDPMP184
Doin Time in Times Square

ADDPMP185
Ballistic Gelatin

ADDPMP186
Mound-building Termites

ADDPMP187
Derby of the Eternal Enemies

ADDPMP188
Episodic Memory
ADDPMP189
Wonderland Amusement Park

ADDPMP190
Water Distribution on Earth
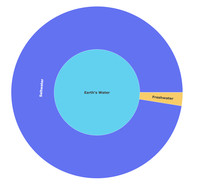
ADDPMP191
Sinkhole

ADDPMP192
The Iceman

ADDPMP193
The Springfield Shopper

ADDPMP194
Ai-Da

ADDPMP195
Ventolin

ADDPMP196
New Car Smell

ADDPMP197
Trepanation

ADDPMP198
Physarum Polycephalum

ADDPMP199
Fugazi, Waiting Room

ADDPMP200
Arpabong
ADDPMP201
Molotov Cocktail

ADDPMP202
Paradise Garage Acoustical Consultation

ADDPMP203
Tamu Massif
ADDPMP204
Luecke Farm

ADDPMP205
MV Blue Marlin

ADDPMP206
Evander Holyfield VS Mike Tyson

ADDPMP207
Smart Gaming Stations

ADDPMP208
Armed Robbery Masks

ADDPMP209
Tanganyika Laughter Epidemic

ADDPMP210
First Item Sold on Ebay

ADDPMP211
Gaboon Viper

ADDPMP212
Sex.com

ADDPMP213
Beipanjiang Bridge

ADDPMP214
The Deadly Art of Survival

ADDPMP215
Lucid Dream

ADDPMP216
Grackles Invade Walmart Parking Lot

ADDPMP217
United States Bullion Depository

ADDPMP218
Dhampir

ADDPMP219
Solar Cooker

ADDPMP220
Antikythera Mechanism
ADDPMP221
Premier League Tattoos

ADDPMP222
Zeusaphone

ADDPMP223
Green Bank Telescope

ADDPMP224
Expo’70 Osaka Demonstration Robot

ADDPMP225
Banggai Cardinalfish

ADDPMP226
Jho Low

ADDPMP227
Bizarre Stock Photos

ADDPMP228
Goûter Refuge

ADDPMP229
Thylacine

ADDPMP230
Abu Dhabi by Jack Burlot

ADDPMP231
Floor Tiling

ADDPMP232
Gold Origins

ADDPMP233
The Neuroscience Of Empathy

ADDPMP234
The Frilled Shark

ADDPMP235
Eye Strain
ADDPMP236
Russian Gangsters Tombstones

ADDPMP237
Tangerine Dream Equipment
ADDPMP238
Garden of Cosmic Speculation

ADDPMP239
Gondola Accident

ADDPMP240
One World In Relation

ADDPMP241
Shuttle Endeavor in the streets of Los Angeles

ADDPMP242
Armillaria Ostoyae

ADDPMP243
Namio Harukawa

ADDPMP244
The Holy Booster

ADDPMP245
Chandra Oppenheim

ADDPMP246
Pangea Proxima
ADDPMP247
Les Apaches

ADDPMP248
Aggressive Mimicry

ADDPMP249
Transocean Winner

ADDPMP250
La Monte Young Piano Piece for Terry Riley #1

ADDPMP251
Catacanthus

ADDPMP252
Tanzanite

ADDPMP253
Chicken Hypnosis

ADDPMP254
Dancing Mania

ADDPMP255
Kalinin K-7

ADDPMP256
Houston Rap Tapes

ADDPMP257
Pentecostal Snake Handlers

ADDPMP258
Instant Housing by Winfried Baumann

ADDPMP259
Maratus Spider

ADDPMP260
James Ensor

ADDPMP261
Sports Bloopers

ADDPMP262
Photo Calibration Targets

ADDPMP263
Claude Young

ADDPMP264
Mano del Desierto

ADDPMP265
Out-of-place Artifact

ADDPMP266
Civil Defense Siren

ADDPMP267
Senna vs Prost Suzuka showdown 1989

ADDPMP268
Husky Dog Talking

ADDPMP269
King David by Ulysses Jenkins

ADDPMP270
Donald Trump-shaped ecstasy pills

ADDPMP271
HiRISE

ADDPMP272
Telescopefish

ADDPMP273
Asperitas

ADDPMP274
Flamethrower

ADDPMP275
Maki Asakawa

ADDPMP276
Halloween by Ken Werner

ADDPMP277
Entoloma Hochstetteri

ADDPMP278
Binoculars Building

ADDPMP279
Photo Finish

ADDPMP280
The Pleasure Shock

ADDPMP281
Fossa dei Leoni

ADDPMP282
French Punk Demo Tapes (1987-1997)

ADDPMP283
Interchanges

ADDPMP284
Ice Hockey Goaltenders Masks

ADDPMP285
The Locked Room

ADDPMP286
Tonic immobility

ADDPMP287
Microsoft Paint Fan Art

ADDPMP288
Form Follows Function

ADDPMP289
Blue

ADDPMP290
Morteza Hannaneh

ADDPMP291
Moulting
ADDPMP292
Lichtenberg Figure

ADDPMP293
Flabellina Iodinea

ADDPMP294
Catwalk Fails

ADDPMP295
Arborglyphs

ADDPMP296
Fluid Dynamics

ADDPMP297
Teatro Del Mundo

ADDPMP298
Macrocilix Maia

ADDPMP299
X-Ray Oddities
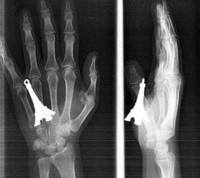
ADDPMP300
Oregon Highway 18 Smiley Face

ADDPMP301
Freezing Rain

ADDPMP302
Blanket Octopus

ADDPMP303
Vandalized Cars

ADDPMP304
Pigeon Photography

ADDPMP305
Prison Landscapes

ADDPMP306
Casa Do Penedo

ADDPMP307
Get Tough
ADDPMP308
Smuggling Schemes

ADDPMP309
Fly Geyser

ADDPMP310
Joseph Jarman

ADDPMP311
Miamira Magnifica

ADDPMP312
Vishapakar

ADDPMP313
Camouflaged Animals

ADDPMP314
Monobloc Chair

ADDPMP315
Paris Syndrome

ADDPMP316
Meltwater Channel

ADDPMP317
Bioluminescent Bacteria

ADDPMP318
Piano Stride

ADDPMP319
Large Hadron Collider
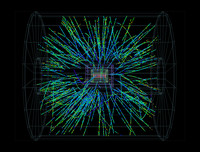
ADDPMP320
The New Ruins of Athens

ADDPMP321
Cool S
ADDPMP322
Syzygy

ADDPMP323
Wolf Collar

ADDPMP324
Aluminium Foiled Rooms

ADDPMP325
Space Debris
ADDPMP326
Dempsey and Firpo

ADDPMP327
Euthanasia Coaster
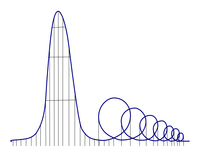
ADDPMP328
Tempescope

ADDPMP329
Sex In Space

ADDPMP330
Lenticular Clouds

ADDPMP331
Body Worlds
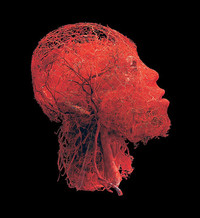
ADDPMP332
House Of Pleasures
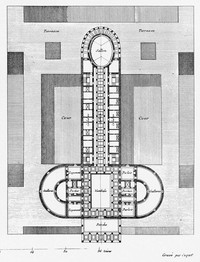
ADDPMP333
Gouldian Finch

ADDPMP334
Vantablack

ADDPMP335
Effects Of Psychoactive Drugs On Spiders

ADDPMP336
Windblown Dogs

ADDPMP337
Timeline Of The Far Future

ADDPMP338
Martin Margiela RTW Spring 1998

ADDPMP339
33 Thomas Street

ADDPMP340
Facial Recognition System

ADDPMP341
Grover Krantz

ADDPMP342
Decimal Time Systems
ADDPMP343
Technophobia

ADDPMP344
Clathrus Ruber

ADDPMP345
Ijen

ADDPMP346
Eiffel Tower Replicas

ADDPMP347
Futurist Cooking

ADDPMP348
Vincenzo Peruggia

ADDPMP349
Bloop
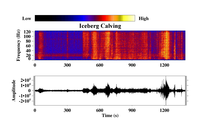
ADDPMP350
Buzkashi

ADDPMP351
Cryptic Firearms

ADDPMP352
Hell Money

ADDPMP353
The Raven Paradox

ADDPMP354
Tract Housing

ADDPMP355
Ricardo López

ADDPMP356
Rag Chair

ADDPMP357
Saddam Hussein's Novels

ADDPMP358
Demon Core

ADDPMP359
Terminator 2 Liquid Metal Effect

ADDPMP360
Odeillo Solar Furnace

ADDPMP361
Pointing Machine

ADDPMP362
Apparent Retrograde Motion
ADDPMP363
Operation Midnight Climax

ADDPMP364
Physalia physalis

ADDPMP365
World's First Photograph

ADDPMP366
Roundabout Public Art

ADDPMP367
Anechoic Chamber

ADDPMP368
Vacuum Effects

ADDPMP369
Stendhal Syndrome

ADDPMP370
Gate Tower Building

ADDPMP371
Out-Of-Body Experience

ADDPMP372
High Fashion By Pawel Jaszczuk

ADDPMP373
Synchronicity
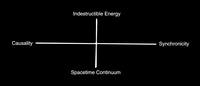
ADDPMP374
Electroencephalophone
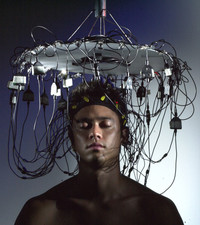
ADDPMP375
Emerald Cockroach Wasp

ADDPMP376
House Gymnastics

ADDPMP377
Rungrado 1st of May Stadium

ADDPMP378
Black Panthers by Agnès Varda

ADDPMP379
Calculator Spelling

ADDPMP380
Embryo Space Colonization
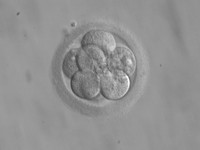
ADDPMP381
Tourist Guy

ADDPMP382
Savonius Wind Turbine

ADDPMP383
Mandrill

ADDPMP384
Autumn Evening

ADDPMP385
Pineal Gland
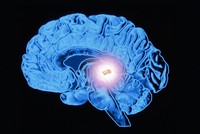
ADDPMP386
Dennis Rodman's Hairstyles

ADDPMP387
Camouflaged Cell Phone Towers

ADDPMP388
Michel Lolito

ADDPMP389
The Jarkov Mammoth

ADDPMP390
The Anarchist Cookbook
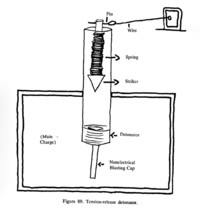
ADDPMP391
carstuckgirls.com

ADDPMP392
Uncanny Valley
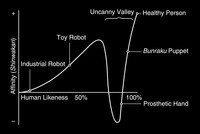
ADDPMP393
Forensic Facial Reconstruction
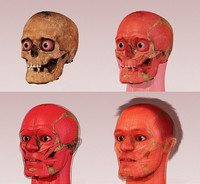
ADDPMP394
Tellus Audio Cassette Magazine

ADDPMP395
Tardigrades In Outer Space
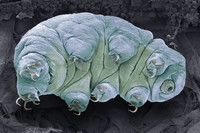
ADDPMP396
The Decline of Western Civilization

ADDPMP397
Parking Space Savers

ADDPMP398
Cosmic Latte

ADDPMP399
Deep Blue

ADDPMP400
Vernacular Architecture

ADDPMP401
Face On Mars
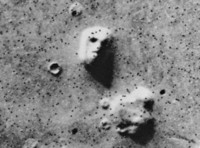
ADDPMP402
Tropical Cyclone Naming

ADDPMP403
Four Color Theorem
ADDPMP404
Fata Morgana

ADDPMP405
Gombe Chimpanzee War

ADDPMP406
Conservation Centers

ADDPMP407
The Philip Experiment

ADDPMP408
Cortical Homunculus
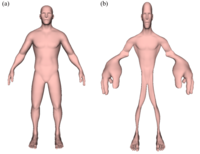
ADDPMP409
Chicxulub Crater

ADDPMP410
Serendipity
ADDPMP411
La Guardia String Figures

ADDPMP412
Bir Tawil
ADDPMP413
Safety Coffin

ADDPMP414
Plant Arithmetic

ADDPMP415
Tank Silencer

ADDPMP416
Parental Advisory

ADDPMP417
Ice House

ADDPMP418
Vanishing Point
ADDPMP419
Celebrity Mugshots

ADDPMP420
Ant Mill

ADDPMP421
Forniphilia

ADDPMP422
Visions of Mars

ADDPMP423
Offshore Architecture

ADDPMP424
Organisms Named After Famous People

ADDPMP425
Kowloon Walled City

ADDPMP426
The Truman Show Delusion

ADDPMP427
Buttered Toast Phenomenon

ADDPMP428
Gates Of Hell

ADDPMP429
Pykrete

ADDPMP430
Zona

ADDPMP431
Perpetual Motion
ADDPMP432
South-Up Map Orientation
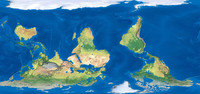
ADDPMP433
Paintings Radiography

ADDPMP434
Five-Hundred-Meter Aperture Spherical Telescope

ADDPMP435
Hiroo Onoda

ADDPMP436
Blue-Ringed Octopus

ADDPMP437
Theremin

ADDPMP438
Premier League Out Of Focus Crowd

ADDPMP439
Cortinarius Violaceus

ADDPMP440
Harbin Ice and Snow Sculpture Festival

ADDPMP441
Olympus Mons
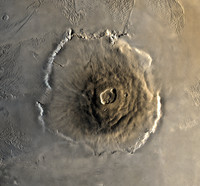
ADDPMP442
Alain Robert

ADDPMP443
Cobalt Blue Tarantula

ADDPMP444
Billboards System Error
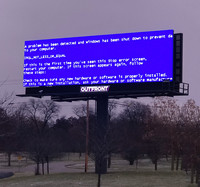
ADDPMP445
Atomic Gardening

ADDPMP446
Wow! Signal
ADDPMP447
Yareta

ADDPMP448
Deja Vu
ADDPMP449
Surface Tension

ADDPMP450
The Richat Structure

ADDPMP451
Deimatic Behaviour

ADDPMP452
High Voltage UV Sparks

ADDPMP453
Horses by Susan Rothenberg

ADDPMP454
Burj Al Babas

ADDPMP455
Cholita Climbers

ADDPMP456
Zoo Hypothesis

ADDPMP457
Pellegrina

ADDPMP458
Heat Haze

ADDPMP459
Impossible Color
ADDPMP460
Zone Of Death
ADDPMP461
Philadelphia Experiment

ADDPMP462
Stubble Burning

ADDPMP463
Other World Kingdom

ADDPMP464
Barcelona Supercomputing Center

ADDPMP465
Xenu

ADDPMP466
Null Island

ADDPMP467
Arachnacris

ADDPMP468
Apeirophobia

ADDPMP469
Volcano Number

ADDPMP470
Cicada 3301

ADDPMP471
Angie Sanclemente Valencia

ADDPMP472
Prohibition Of Dying

ADDPMP473
Elephant's Toothpaste

ADDPMP474
Turner Doomsday Video

ADDPMP475
As Slow As Possible

ADDPMP476
Pieing

ADDPMP477
Archaeoacoustics

ADDPMP478
Metta World Peace

ADDPMP479
Titanosauria

ADDPMP480
Pareidolia

ADDPMP481
Animal Chimera

ADDPMP482
Brain Uploading
ADDPMP483
Hellmouth

ADDPMP484
China National Highway 110 traffic jam

ADDPMP485
Eigenface

ADDPMP486
Miss Belvedere

ADDPMP487
Moving Sofa Problem
ADDPMP488
Ichthyoallyeinotoxism

ADDPMP489
Spaghettification

ADDPMP490
There Are Known Knowns

ADDPMP491
Westbury White Horse

ADDPMP492
Expanding Earth
ADDPMP493
X-Seed 4000

ADDPMP494
List of films that most frequently use the word "fuck"

ADDPMP495
Conspiracy 58

ADDPMP496
Mamuthones Pagan Ritual

ADDPMP497
Geologic Calendar

ADDPMP498
Antarctica Blood Falls

ADDPMP499
Dream Argument

ADDPMP500
Big Dumb Object

ADDPMP501
Snowball Earth

ADDPMP502
Colorless Green Ideas Sleep Furiously
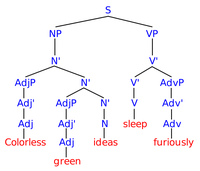
ADDPMP503
Ophiocordyceps unilateralis

ADDPMP504
Macdonald Triad

ADDPMP505
Mir Mine

ADDPMP506
Pinhead Design

ADDPMP507
Multiverse

ADDPMP508
Tetris Effect

ADDPMP509
Future Bugs

ADDPMP510
Saturn's Hexagone

ADDPMP511
Controlled Impact Demonstration

ADDPMP512
Maltese Tiger

ADDPMP513
Legislative Violence

ADDPMP514
Blast Wave

ADDPMP515
Pallasite
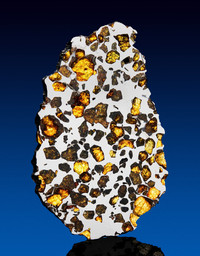
ADDPMP516
Plan Voisin

ADDPMP517
Ideasthesia

ADDPMP518
Eunice Aphroditois

ADDPMP519
Tornado Alley

ADDPMP520
Trading Rooms

ADDPMP521
Peanut Hole
ADDPMP522
Schmidt Sting Pain Index

ADDPMP523
Greater Green River Intergalactic Spaceport

ADDPMP524
Obetrol

ADDPMP525
Articulatory Phonetics
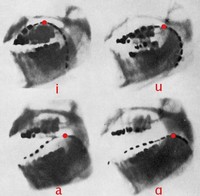
ADDPMP526
Googol
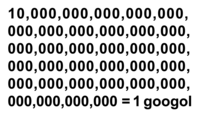
ADDPMP527
Dizzy Gillespie 1964 Presidential Campaign

ADDPMP528
Tidal Bore

ADDPMP529
Longyangxia Dam

ADDPMP530
Winona Ryder Shoplifting

ADDPMP531
Thagomizer

ADDPMP532
Exploding Head Syndrome

ADDPMP533
Dreamachine

ADDPMP534
Birthday Effect

ADDPMP535
Bananadine

ADDPMP536
Coevolution

ADDPMP537
Battle of Surfaces

ADDPMP538
Vladimir Demikhov

ADDPMP539
844-NEED-SCI

ADDPMP540
Tabular Iceberg

ADDPMP541
Chindōgu

ADDPMP542
Photovoltaic Panels

ADDPMP543
Ribs Recordings
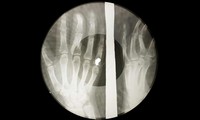
ADDPMP544
Kirov Reservoir

ADDPMP545
Lüscher Color Test
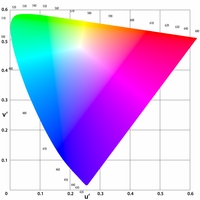
ADDPMP546
Busta Rhymes Island
ADDPMP547
Fires of Kuwait

ADDPMP548
Bite Suit Fashion

ADDPMP549
Hyperart Thomasson

ADDPMP550
Coloboma
ADDPMP551
Plants In Space

ADDPMP552
Pyramid Scheme
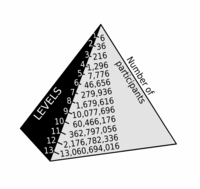
ADDPMP553
Here And Elsewhere

ADDPMP554
Loudest Sound Ever Recorded

ADDPMP555
Phineas Gage
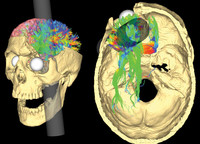
ADDPMP556
Klencke Atlas

ADDPMP557
Centennial Light

ADDPMP558
Fallstreak Hole

ADDPMP559
Animal Prosthetics
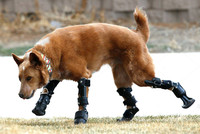
ADDPMP560
Catatumbo Lightning

ADDPMP561
Yaeba

ADDPMP562
Greenwashing

ADDPMP563
FBI Ten Most Wanted Fugitives

ADDPMP564
The Mind's Eye

ADDPMP565
Agalmatophilia

ADDPMP566
Cerro Torre Controversy

ADDPMP567
Antimatter Cost
ADDPMP568
Weekly World News Recurring Subjects

ADDPMP569
Monster Trucks Construction

ADDPMP570
La Guardia Committee

ADDPMP571
Hawk-Eye Technology

ADDPMP572
Afterimage

ADDPMP573
Animals by Number of Neurons

ADDPMP574
Earthquake Baroque

ADDPMP575
Water Overdose

ADDPMP576
Johnson's Criteria

ADDPMP577
Yellow

ADDPMP578
Tilikum

ADDPMP579
Aerican Empire

ADDPMP580
Junko Tabei

ADDPMP581
Archer’s Paradox

ADDPMP582
Animalia Paradoxa

ADDPMP583
Symphony of the Seas

ADDPMP584
Ehlers–Danlos Syndromes
ADDPMP585
Bridge to Nowhere

ADDPMP586
Lissajous Curve
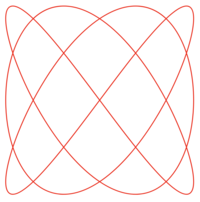
ADDPMP587
Vortex Project

ADDPMP588
Ryuichi Sakamoto OST Credits

ADDPMP589
Blue

ADDPMP590
Defense Architecture

ADDPMP591
Prehistoric Warfare

ADDPMP592
Immortal Jellyfish

ADDPMP593
House Relocation

ADDPMP594
Extelligence
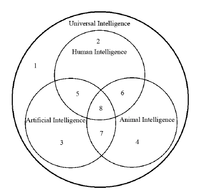
ADDPMP595
Aircraft Camouflage

ADDPMP596
Parkett Magazine

ADDPMP597
Ferrari World Abu Dhabi

ADDPMP598
D.C. Sniper Attacks

ADDPMP599
Memristor

ADDPMP600
Icy blast in Sahara

ADDPMP601
The blind leading the blind

ADDPMP602
Poetry on the Runway

ADDPMP603
Eurasian hoopoe

ADDPMP604
Three-card Monte

ADDPMP605
Yakhchāl

ADDPMP606
Mensa International

ADDPMP607
Through The Looking Glass

ADDPMP608
F-Pattern
ADDPMP609
Selenicereus grandiflorus

ADDPMP610
La Tartuguiere

ADDPMP611
We Are Our Mountains

ADDPMP612
Courtroom Sketches

ADDPMP613
Chūken Hachikō

ADDPMP614
Devil's Horn Solar Eclipse
ADDPMP615
Wim Hof

ADDPMP616
DrugsData

ADDPMP617
Asaro Mudmen

ADDPMP618
Faint To Attention

ADDPMP619
Smoke Signal

ADDPMP620
O.S.T. Credits

ADDPMP621
Ostehøvel

ADDPMP622
Green flash

ADDPMP623
Eugen Sandow

ADDPMP624
449 Hamburga

ADDPMP625
Fantasy coffin

ADDPMP626
First text

ADDPMP627
Eminem with fans

ADDPMP628
The Ancient Order of Druids

ADDPMP629
The Earth Room

ADDPMP630
Goose bumps

ADDPMP631
Breviceps fuscus

ADDPMP632
Cloud weight

ADDPMP633
Wheatfield — A Confrontation

ADDPMP634
Heterochromia iridum

ADDPMP635
Barnard's universal cipher code
ADDPMP636
Carcinisation

ADDPMP637
Felt cloak

ADDPMP638
Sarco pod

ADDPMP639
Rap Snacks

ADDPMP640
William Charles Utermohlen

ADDPMP641
ILOVEYOU

ADDPMP642
Jacksonville Turbine Interchange

ADDPMP643
Hardiman

ADDPMP644
Bell-mouth spillway

ADDPMP645
Rest on the Flight into Egypt

ADDPMP646
Oosterscheldekering

ADDPMP647
Unsolved Mysteries

ADDPMP648
Celebrities looking like statues

ADDPMP649
Red-lipped batfish

ADDPMP650
Tokyo Olympiad

ADDPMP651
Tunnel vision

ADDPMP652
Cocaine Bear

ADDPMP653
Leonids

ADDPMP654
Our Lady of Medjugorje

ADDPMP655
Brain Computer Interface
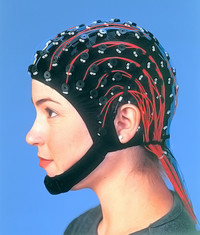
ADDPMP656
Robert Pershing Wadlow

ADDPMP657
Horse Clipping Styles

ADDPMP658
Fictional Characters On Set

ADDPMP659
CUE

ADDPMP660
James Howells

ADDPMP661
Cloud cover

ADDPMP662
Sunset on Mars

ADDPMP663
Paleontologists Posing

ADDPMP664
DoggoLingo

ADDPMP665
The Large Cloth of Abuse

ADDPMP666
Shi Pei Pu

ADDPMP667
Tiger beetle

ADDPMP668
Chaos theory
ADDPMP669
Mimetic Hot Air Balloons

ADDPMP670
Statue Of Liberty Centennial Restoration

ADDPMP671
Frame
ADDPMP672
Polar Stations Architecture

ADDPMP673
Spray-Painted Sheep

ADDPMP674
Tephra

ADDPMP675
Novelty Architecture

ADDPMP676
Ojos del Salado

ADDPMP677
Bez

ADDPMP679
The Turin Shroud

ADDPMP680
Birds aren't real

ADDPMP681
Beaux-Arts Ball

ADDPMP682
Celebrities Hidding

ADDPMP683
The Hand

ADDPMP684
Pavarotti in Hyde Park

ADDPMP685
Prada Counterfeit
ADDPMP686
Stroboscopic Effect

ADDPMP687
Sprite Lightning
ADDPMP688
Theory of Everything
ADDPMP689
Krampus

ADDPMP690
1933 Double Eagle

ADDPMP691
Sylacauga

ADDPMP692
Circular Error Probable

ADDPMP693
Stygian Owl

ADDPMP694
Charles Darwin Office Chair

ADDPMP695
Nefertiti Missing Eye

ADDPMP696
Heat Influence

ADDPMP697
Dai Heiwa Kinen To

ADDPMP698
Raymond Robinson

ADDPMP699
Persistence of Vision
ADDPMP700
Diplomacy Design

ADDPMP701
Atlanta Nights

ADDPMP702
Frogman

ADDPMP703
Rashomon Effect

ADDPMP704
Biomusic

ADDPMP705
Adaptive Colouration in Animals

ADDPMP706
Knowledge Ark

ADDPMP707
Pierre Brassau

ADDPMP708
Byrne, Glass and Ginsberg on Arthur Russell 'Another Thought'

ADDPMP709
Artificial Cranial Deformation
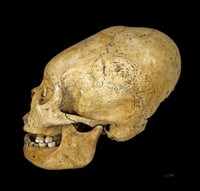
ADDPMP710
Coccoloba Gigantifolia

ADDPMP711
Ouroboros
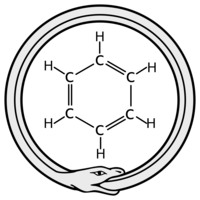
ADDPMP712
Ekman 60 Faces Task

ADDPMP713
Monument to Humanity

ADDPMP714
Percussive Maintenance

ADDPMP715
Mass Wasting

ADDPMP716
Digital Dystopia

ADDPMP717
Megalodon, The Monster Shark Lives
ADDPMP718
Perp Walk

ADDPMP719
Index Librorum Prohibitorum

ADDPMP720
Hellas Planitia

ADDPMP721
Contour Ploughing

ADDPMP722
Tarrare

ADDPMP723
Green Night Vision

ADDPMP724
Funktion-One

ADDPMP725
Machapuchare

ADDPMP726
Arnold Schwarzenegger at the Whitney Museum

ADDPMP727
Selective Amnesia
ADDPMP728
Hakken

ADDPMP729
Hypertrichosis

ADDPMP730
Wilhelm Loeser

ADDPMP731
Devil's Advocate

ADDPMP732
Mandible
ADDPMP733
Gecko's Feet

ADDPMP734
US Flag Desecration

ADDPMP735
Zero Minutes Zero Seconds

ADDPMP736
Illegal Names

ADDPMP737
LSD: Dream Emulator

ADDPMP738
Harkonnen Chair
ADDPMP739
The Orwell Leg

ADDPMP740
Miroslav Tichý

ADDPMP741
Bird-of-Paradise

ADDPMP742
Phos-Chek

ADDPMP743
Ghosts... of the Civil Dead

ADDPMP744
Hyperbolic Paraboloid

ADDPMP745
Morning Glory Cloud

ADDPMP746
Mongolian Death Worm

ADDPMP747
Forensic Engineering

ADDPMP748
George Gurdjieff

ADDPMP749
Hostile Architecture

ADDPMP750
Championship Belt

ADDPMP751
Hollow-Face Illusion

ADDPMP752
Doomsday Clock

ADDPMP753
Elizabethan Collar

ADDPMP754
Skip Zone

ADDPMP755
Ghost Detainee

ADDPMP756
Society for the Preservation of the Quazer Beast
ADDPMP757
Shock Diamond

ADDPMP758
Newfoundland Icebergs

ADDPMP759
Moreton Wave
ADDPMP760
Pelorus Jack

ADDPMP761
Space Selfie

ADDPMP762
Six Degrees of Freedom

ADDPMP763
Sutton Hoo helmet

ADDPMP764
Acámbaro Figures

ADDPMP765
Liberty Spikes

ADDPMP766
Aorounga

ADDPMP767
Backboard Shattering

ADDPMP768
Bite Force Quotient

ADDPMP769
Robert Cailliau

ADDPMP770
New Atlantis

ADDPMP771
Hexakosioihexekontahexaphobia

ADDPMP772
Shimizu Mega-City

ADDPMP773
Information-Theoretic Death

ADDPMP774
Frat House

ADDPMP775
Cullinan Diamond

ADDPMP776
Intempo

ADDPMP777
Pseudocreobotra Wahlbergii

ADDPMP778
Fifa Evolution

ADDPMP779
Amazonian Royal Flycatcher

ADDPMP780
First Ever X-ray
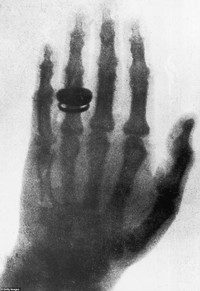
ADDPMP781
Behaviour-Altering Parasite
ADDPMP782
Wind Swept Trees

ADDPMP783
The Falling Man

ADDPMP784
Riace Bronzes

ADDPMP785
Carbon Chauvinism

ADDPMP786
Hunga Tonga–Hunga Haʻapai

ADDPMP787
Spion Kop

ADDPMP788
Forensic Arts

ADDPMP789
Drogue Parachute

ADDPMP790
Shabono

ADDPMP791
Lithophone

ADDPMP792
Paranoiac-Critical Method

ADDPMP793
Kubotan

ADDPMP794
Meteorite Pricing

ADDPMP795
Lincos

ADDPMP796
Walking Truck

ADDPMP797
Moses Bridge

ADDPMP798
Greebles

ADDPMP799
Secchi Disc

ADDPMP800
Pan-American Highway

ADDPMP801
Myostatin-related Animal Hypertrophy

ADDPMP802
Metal Umlaut

ADDPMP803
Enantiodromia

ADDPMP804
Fire Whirl

ADDPMP805
Pupil Patterns

ADDPMP806
Biotic Baking Brigade

ADDPMP807
Baghdad Battery

ADDPMP808
Makapansgat Pebble
ADDPMP809
Zombie Taxon

ADDPMP810
Google Earth Misuse
