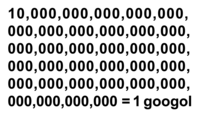Baghdad Battery
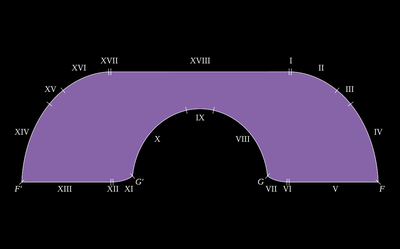
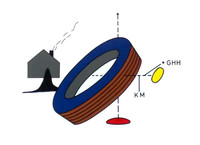
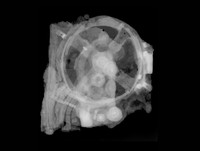
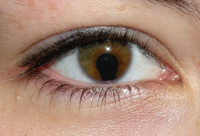
ADDPMP807The artefacts consist of a terracotta pot approximately 140mm (6in) tall with a 38mm (1.5in) mouth containing a cylinder made of a rolled copper sheet which houses a single iron rod. At the top, the iron rod is isolated from the copper by bitumen with plugs or stoppers and both rod and cylinder fit snugly inside the opening of the jar. The copper cylinder is not watertight so if the jar were filled with a liquid, this would surround the iron rod as well. The artefact has been exposed to the weather and has suffered corrosion.
German archaeologist Wilhelm König, at the time director of the National Museum of Iraq, thought the objects might date to the Parthian period, between 250BC and AD224 but this remains unclear, as does their purpose. König observed a number of very fine silver objects from ancient Iraq plated with very thin layers of gold and speculated that they were electroplated. In 1938, he authored a paper offering the hypothesis that they may have formed a galvanic cell, perhaps used for electroplating gold onto silver objects. Also, the corrosion of the metal and tests both indicated that an acidic agent such as wine or vinegar was present in the jar. This led to speculation that the liquid was used as an acidic electrolyte solution to generate an electric current from the difference between the electrode potentials of the copper and iron electrodes. Though the iron rod did project outside of the asphalt plug, the copper tube did not, making it impossible to connect a wire to this to complete a circuit. Consequently, the interpretation of the Baghdad battery as an electroplated object is rejected by sceptics.